Induced asthma – New HDM (immuno)therapy testing (45 days)
Model 5: New HDM (immuno)therapy testing (46 days)
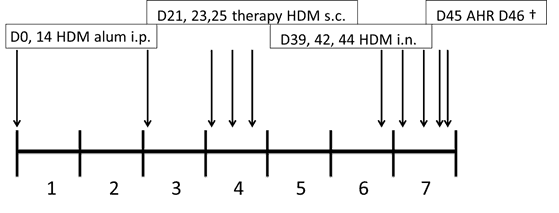
- Description: Sensitized with HDM by intranasal administration on days 0 and 14 followed by SCIT therapy on days 21, 23 and 25, followed by intranasal challenge at days 39, 42 and 44, performing an AHR test on day 45 and sacrificing mice at day 46.
- Duration: 46 days
- Attributes: New HDM (Immuno) therapy testing
- Endpoints: Airway hyper-reactivity and detailed lung mechanics; flexiVent™. Eosinophil cell count in the BALF. Inflammatory mediator content.
- Material used: D. pteronyssinus extract from Citeq
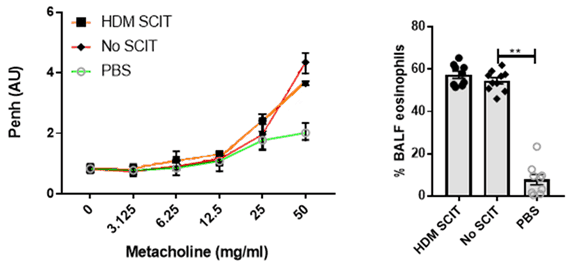
HDM SCIT induces an increase in airway resistance and an eosinophilic response.
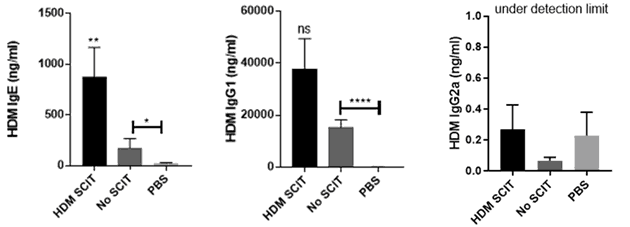
HDM SCIT induces a higher number of HDM IgE and IgG1 compared to No SCIT and PBS treated mice. Key hallmarks of HDM induces allergic asthma are present when Citeq HDM extract is used.
