What are Endotoxins (LPS)
Endotoxin, also referred to as Lipopolysacharides (LPS), is the major component of the outer membrane of Gram-negative bacteria and is released upon disintegration of the intact bacteria (Gorbet et al, 2004). This can occur either through growth, division, cell death, lyses or mechanical destruction during the manufacturing process. The endotoxins comprise of a core polysaccharide chain, O-specific polysaccharide side chains (O-antigen) and a lipid component, Lipid A. Lipid A is responsible for the toxic effect of the endotoxins. The function of endotoxins in a gram negative bacteria is mainly protective and structural.
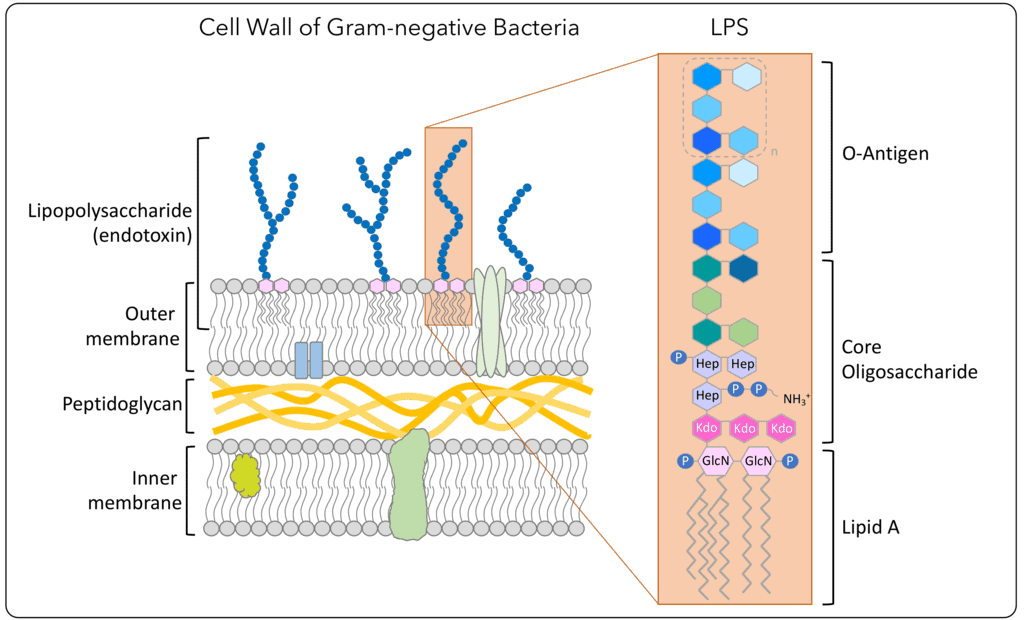
History of endotoxins
Endotoxins were discovered by Richard Friedrich Johannes Pfeiffer. He named them endotoxins, in opposition with exotoxins, which are toxins that are released by bacteria into the surrounding environment. In his opinion the endotoxins could release their toxins only after the cell wall of the bacteria was destroyed (which was proven later that is not necessarily true). Examination of endotoxins is necessary for microbial and animal cell-culture operations that produce active pharmaceutical ingredients (APIs) and vaccines. If a cell culture is contaminated with endotoxins, it could affect cell growth and function (thus potentially affecting product quality). Biopharmaceutical companies need to understand the endotoxin levels within their operations to gain more understanding of working cells. In this article, BioProcess International’s senior technical editor Cheryl Scott discusses endotoxin monitoring and control with Lisa Komski from Fujifilm Wako Chemicals USA Corporation.
Low endotoxin HDM extract
Citeq cultivates house dust mites (HDM) such as D. pteronyssinus and D. farinae and manufactures high quality HDM products that can be used for diagnostic and research applications. HDM extracts are one the main products of Citeq and they are composed of allergens, non-allergens and endotoxins. Endotoxins, also referred to as Lipopolysacharides (LPS), can affect cell growth and function (thus potentially affecting product quality) and therefor examination of endotoxins is necessary. We have developed an endotoxin low D. pteronyssinus extract and D. farinae extract. The endotoxins are removed during the manufacturing process which is carefully monitored by routine testing with an endotoxin assay from Lonza (PyroGeneTM Recombinant Factor C, range: 0.005-5 EU/mL). HDM extracts and endotoxin low HDM extracts differ in their endotoxin content, however both contain the same amount of major allergens (Table 1). Table 1: Comparison between regular HDM extracts and endotoxin low HDM extracts from Citeq.
| Specifications | HDM extracts (D. pteronyssinus/ D. farinae) | Endotoxin low HDM extracts (D. pteronyssinus/ D. farinae) | |
| Group 1 major allergens (mg/g) | 5 – 100 | 1 – 25 | |
| Group 2 major allergens (mg/g) | 0.5 – 10 | 0.1 – 5.5 | |
| Protein content (mg/g) | 25 -250 | 3-75 (brad) 70-350 (BCA) | |
| Endotoxin (EU/mg) | 500 – 7500 | 1 – 50 (DP) / 300-750 (DF) | |
| Protease activity | Serine (mU/mg) | 103 | TBD |
| Cysteine (U/mg) | 104 | TBD | |
| Mycoplasma DNA presence | Negative | Negative | |
Why are there bacteria in house dust mites and endotoxins in HDM extracts?
Evolutionary speaking, the link between bacteria and house dust mites should not be surprising. It is long hypothesized that the ancestors of modern house dust mites were living as permanent parasites on birds and mammals whereas house dust mites today live as commensals in vertebrate nests and houses (Hubert et al, 2019). This change of habitat required adaptations the living temperature, humidity and the ability to digest dander, hair and nails. Such food sources are composed of collagen, elastin and keratin which are difficult to digest. Very few animals are able to digest keratin and interaction between bacteria and house dust mites could explain their ability of feeding on dander, hair and nails. Several studies have shown that house dust mites have a microbiome composed of several bacteria and fungi. The following species of bacteria have been identified in house dust mites: Bartonella, Cardinium, Caulobacter, Chitinophagaceae (All gram-), Kocuria and Lactobacillus (Both gram+). Fungi species Aspergillus, Candida, Saccharomyces and Wallemia were also shown (Hubert et al, 2019; Kopeky et al, 2014; Molva et al, 2020). House dust mites and the presence of bacteria thus go hand in hand, it is therefore common that HDM extracts contain endotoxins.
Effect of endotoxins in cell culture experiments
House dust mite extracts are widely used for diagnostic, immunotherapy and research purposes. They are composed of a wide variety of molecules including: allergens, non-allergens and endotoxins. Either of these components can have a different effect skin, lung and immune cells which is important to realize when conducting cell culture experiments with HDM extracts. Molecules in the HDM extract can affect the cells through activation or suppression of receptors molecules that are located on the cell membrane. Many cells have Toll-Like-Receptors (TLRs) on their cell membrane which plays a role in the innate immune response. TLRs are primarily expressed on dendritic cells, macrophages, epithelial cells and fibroblast and recognize pathogen-associated molecular patterns (Kawasaki et al, 2014). It has been shown that endotoxins are able initiate an innate immune response through the activation of TLR2, TRL4, CD14 and MD-2 (Rockwood et al, 2013). Endotoxins can affect cell growth, function and in vitro fertilization at concentrations from > 0.1 ng/mL (Nomura et al, 2017; Fishel et al, 1988). HDM allergens, such as Der p1 and Der p5, also initiate an innate immune response however through activation of Protease Activated Receptor-1 (PAR-1) and PAR-2 (Kaufmann et al, 2006). So in summary, it is important to be aware of the different components in HDM extracts that can initiate an inflammatory response in cells through different receptors (Table 2). And depending on your experiment and cell type you should consider to investigate a HDM extract, HDM extract with low endotoxin or both. Table 2: Overview of the effects of endotoxin and HDM extracts in various cell lines.
| Cell line | Experimental setup | Dose | Effect | Reference |
| ENDOTOXINS | ||||
| Human alveolar macrophages | Incubation with different concentrations of E. coli endotoxin for 1 and 24h | 1, 10 and 100 µg/mL | Exposure > 10 µg/mL endotoxin was cytotoxic and inhibited phagocytosis, adherence and spreading | Davis et al, 1980 |
| Human mesenchymal stem cells | Cultivation for 40 days in the prescence of various concentrations endotoxin | 0, 0.1, 1, 10, 100 and 1000 ng/ml | Endotoxin exposure > 0.1 ng/mL has adverse effects on MSC proliferation | Nomura et al, 2017 |
| Human in vitro fertilization | In vitro fertilization in the presence of endotoxins | > 0.1 ng/mL or < 0.1 ng/mL | Decreased incidence of pregnancy in the presence of > 0.1 ng/mL endotoxin | Fishel et al, 1988 |
| ALLERGENS | ||||
| Human basophils (KU812) | KU812 cells were incubated for 2 hours with native Der p1 KU812 cells were incubated for 24 hours with Der p1 | 1 µg/mL | Der p1 exposure for 2h induces expression of IL-4, IL-5 and IL-13 mRNA Der p1 exposure for 24h stimulates production of IL-4 and IL-13 cytokines | Phillips et al, 2003 |
| A-549 (human alveolar type II pneumocyte) | A-549 cells were incubated for 17 hours with native Der p1 | 0 – 250 nM | Der p1 exposure for 17h dose-dependently increased IL-8 secretion | Adam et al, 2006 |
| HeLa cells (human cervical cancer cell) | HeLa cells were incubated for 7 hours with native Der p1 | 150 nM | Der p1 exposure for 7h increased IL-8 secretion | |
| HDM EXTRACTS WITH AND WITHOUT ENDOTOXINS | ||||
| HMVEC-D (adult dermal microvascular endothelial cells) | Cells were stimulated for 6, 12 and 24 hours with varying doses of DP and DF HDM extract containing endotoxin Cells were stimulated for 6 hours with DP and DF HDM extract containing polymixin B inactivated-endotoxin | 0, 25 , 50 and 100 µg/mL 100 µg/mL | Exposure to HDM extract with endotoxin dose- and time-dependently increased expression of: ICAM-1 and VCAM-1. E-selectin expression was also increased In addition exposure induced dose- and time-dependent secretion of IL-6, IL-8, MCP-1, and GM-CSF Exposure to HDM extracts containing inactivated-endotoxins results in normalized expression of ICAM-1, VCAM-1, and E-selectin and showed little or no secretion of IL-6, G-CSF, and GM-CSF | Arlian et al, 2009 |
| Differentiated primary bronchial epithelial cells | Cells were stimulated for 24 and 48 hours with endotoxin low HDM extract from Citeq | 100 and 200 µg/mL | Endotoxin content in the HDM extract is acceptable and does not significantly influence immune responses in the tested experimental set up. | Customer data, 2020 |
Advantages of endotoxin low HDM extracts
Citeq manufactures HDM extracts that are low in endotoxin for both Dermatophagoides pteronyssinus and D. farinae that are the most common species of house dust mites. The manufacturing process for these products is directed towards a minimal exposure to environmental microorganisms/ endotoxins and a maximal removal of house dust mite associated endotoxins (Citeq, proprietary data). Another advantage is that the endotoxins low extracts are lyophilized in sterile flasks at prefilled manageable quantities, which provides you with an easy to use product in your research model. As stated in the previous paragraph, endotoxin low HDM extracts are interesting option to either validate your research model or investigate the role of endotoxins in the development of HDM allergies/immune responses. For more information and questions, please contact us via the contact form or via info@citeq.com.
References
- Adam E, Hansen KK, Astudillo Fernandez O, Coulon L, Bex F, Duhant X, Jaumotte E, Hollenberg MD, Jacquet A. The house dust mite allergen Der p 1, unlike Der p 3, stimulates the expression of interleukin-8 in human airway epithelial cells via a proteinase-activated receptor-2-independent mechanism. J Biol Chem. 2006 Mar 17;281(11):6910-23.
- Arlian LG, Elder BL, Morgan MS. House dust mite extracts activate cultured human dermal endothelial cells to express adhesion molecules and secrete cytokines. J Med Entomol. 2009 May;46(3):595-604.
- Davis WB, Barsoum IS, Ramwell PW, Yeager H Jr. Human alveolar macrophages: effects of endotoxin in vitro. Infect Immun. 1980 Dec;30(3):753-8.
- Fishel S, Jackson P, Webster J, Faratian B. Endotoxins in culture medium for human in vitro fertilization. Fertil Steril. 1988 Jan;49(1):108-11.
- Gorbet MB, Sefton MV. Biomaterial-associated thrombosis: roles of coagulation factors, complement, platelets and leukocytes. Biomaterials. 2004 Nov;25(26):5681-703.
- Hubert J, Nesvorna M, Kopecky J, Erban T, Klimov P. Population and Culture Age Influence the Microbiome Profiles of House Dust Mites. Microb Ecol. 2019 May;77(4):1048-1066.
- Kauffman HF, Tamm M, Timmerman JA, Borger P. House dust mite major allergens Der p 1 and Der p 5 activate human airway-derived epithelial cells by protease- dependent and protease-independent mechanisms. Clin Mol Allergy. 2006 Mar 28;4:5.
- Kawasaki T, Kawai T. Toll-like receptor signaling pathways. Front Immunol. 2014 Sep 25;5:461.
- Kopecký J, Nesvorná M, Hubert J. Bartonella-like bacteria carried by domestic mite species. Exp Appl Acarol. 2014;64(1):21-32.
- Molva V, Bostlova M, Nesvorna M, Hubert J. Do the microorganisms from laboratory culture spent growth medium affect house dust mite fitness and microbiome composition? Insect Sci. 2020 Apr;27(2):266-275.
- Nomura Y, Fukui C, Morishita Y, Haishima Y. A biological study establishing the endotoxin limit for in vitro proliferation of human mesenchymal stem cells. Regen Ther. 2017 Sep 9;7:45-51.
- Phillips C, Coward WR, Pritchard DI, Hewitt CR. Basophils express a type 2 cytokine profile on exposure to proteases from helminths and house dust mites. J Leukoc Biol. 2003 Jan;73(1):165-71.
- Rockwood J, Morgan MS, Arlian LG. Proteins and endotoxin in house dust mite extracts modulate cytokine secretion and gene expression by dermal fibroblasts. Exp Appl Acarol. 2013 Nov;61(3):311-25.
